The U.S. Food and Drug Administration’s accelerated approval program is meant to give patients early access to promising drugs. But how often do these drugs actually improve or extend patients’ lives?
In a new study, researchers found that most cancer drugs granted accelerated approval do not demonstrate such benefits within five years.
“Five years after the initial accelerated approval, you should have a definitive answer,” said Dr. Ezekiel Emanuel, a cancer specialist and bioethicist at the University of Pennsylvania who was not involved in the research. “Thousands of people are getting those drugs. That seems a mistake if we don’t know whether they work or not.”
The program was created in 1992 to speed access to HIV drugs. Today, 85% of accelerated approvals go to cancer drugs.
It allows the FDA to grant early approval to drugs that show promising initial results for treating debilitating or fatal diseases. In exchange, drug companies are expected to do rigorous testing and produce better evidence before gaining full approval.
 Nuggets blow 20
Nuggets blow 20 King Charles leaves hospital as Kate recovers at home
King Charles leaves hospital as Kate recovers at homeAmtrak train hits pickup truck in upstate New York, 3 dead including child
 NEW YORK (AP) — A child was among the three victims killed when a passenger train hit a pickup truck ...[Detailed]
NEW YORK (AP) — A child was among the three victims killed when a passenger train hit a pickup truck ...[Detailed]Supermarket facial recognition trial: Rotorua mother’s ‘discrimination’ ordeal
 By Sandra Conchie of ...[Detailed]
By Sandra Conchie of ...[Detailed]Government thinks disabled 'add no value to society'
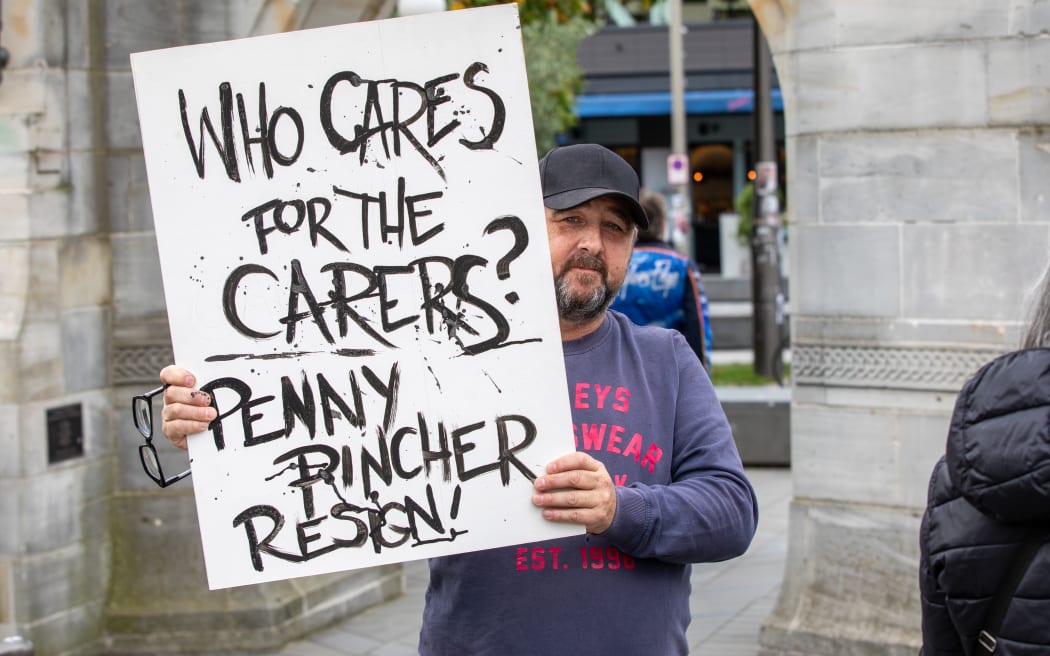 People who attended a rally at the Bridge of Remembrance in Christchurch on Thursday. Photo: RNZ / N ...[Detailed]
People who attended a rally at the Bridge of Remembrance in Christchurch on Thursday. Photo: RNZ / N ...[Detailed]Joe Biden Inauguration: World hopes for renewed cooperation with US under Biden
 Your web browser is no longer supported. To improve your experience update it here ...[Detailed]
Your web browser is no longer supported. To improve your experience update it here ...[Detailed]Travis Kelce downs whiskey shot on slice of bread at Kelce Jam without Taylor Swift
More toll roads coming, but at what price?
 Photo: NZTA ...[Detailed]
Photo: NZTA ...[Detailed]Farmers selling stock, land as 'extreme' drought dries up streams
 By Georgie HanafinAwatere Valley (file image). Photo: RNZ/Sally Round ...[Detailed]
By Georgie HanafinAwatere Valley (file image). Photo: RNZ/Sally Round ...[Detailed]China crackdown on Tiananmen Square commemorations extends to Hong Kong
 Your web browser is no longer supported. To improve your experience update it here ...[Detailed]
Your web browser is no longer supported. To improve your experience update it here ...[Detailed]Verona confirms Serie A status for another year after beating Salernitana
 SALERNO, Italy (AP) — Verona secured a place in next year’s Serie A after beating Salernitana 2-1 on ...[Detailed]
SALERNO, Italy (AP) — Verona secured a place in next year’s Serie A after beating Salernitana 2-1 on ...[Detailed] Your web browser is no longer supported. To improve your experience update it here ...[Detailed]
Your web browser is no longer supported. To improve your experience update it here ...[Detailed]Supreme Court rejects an appeal from a Canadian man once held at Guantanamo

Māori communities must be at forefront of emergency management plans, hapū say
